COMPANY
Yunsung F&C, growing based on quality and technology.
Quality Management Program
Always striving to achieve customer satisfaction through the best quality.
Quality Management Policy
 Please move the screen left and right.
Please move the screen left and right.

Quality Management System
-
Policy
Top Quality Management
Direction for
Quality Assurance
-
Quality Manual
YS QAM-100 : ISO9001 : 2008
YS QAM-200 : ASME U
YS QCM-300 : CSEL
-
Quality
ProcedureYS QAP-1000
: Quality Assurance Procedure
-
Documentation
Quality Verification Data
Design Related Records
Achieving customer satisfaction through the best quality service
Quality Management Flowchart
 Please move the screen left and right.
Please move the screen left and right.

Design Quality Control
-
STEP 01
Project Initiation
- Establishment of Design Execution Plan
SPEC. Review
-
Project Project Initiation Result
Project K. O. M
-
STEP 02
Establishment of Design Concept
- Approval/Regulation Review Document
SPEC. Review
-
Establishment of Design Policy
Design Meeting
-
STEP 03
Detail Design (FA stage)
- Technical Review
Preliminary Order Documents
Approval Documents
-
Preparation ofFA Documents
Quality Check (1st round)
-
STEP 04
Detail Design (AFC stage)
- AFC Documents
B/M Outputs Specification Documents
Customer Requirements
-
Preparation of AFC Documents
Quality Check (2nd round)
-
STEP 05
Drawing Changes (Revision & F/U stage)
- Revised Documents Reflection of Customer Feedback
-
Quality Issue Documents
Quality Meeting
-
STEP 06
Drawing Changes (As-built stage)
- As-built Documents Approval Documents
-
Submission of Project Outcome
Submission of T/O Document
-
STEP 07
Project Close Out
- Final Document Submission
V-Model Approach for Equipment Suitability Evaluation.
The V-model is primarily used to manage the validation sequence for manufacturing and support equipment such as vessels.
In the V model, the documents for DQ (design qualification) and the protocols for IQ (installation qualification), OQ (operation qualification),and PQ (performance qualification) are linked together. Commissioning
 Please move the screen left and right.
Please move the screen left and right.

Suitability Qualification of Vessels
Vessel types have a direct impact on products, and some vessels play an auxiliary role in the process. The decision to conduct only a commissioning testor to perform a suitability evaluation depends on its intended use. Risk assessment can be used to make such decisions.
- Solution formulation/mixing vessels
- Synthetic vessels
- Injection solution
- IBC/Blender
- Bio and culture vessels
- Purified water/distilled water storage tanks
Representatively, the types of vessels that require Suitability Qualification evaluation are as follows
 Please move the screen left and right.
Please move the screen left and right.

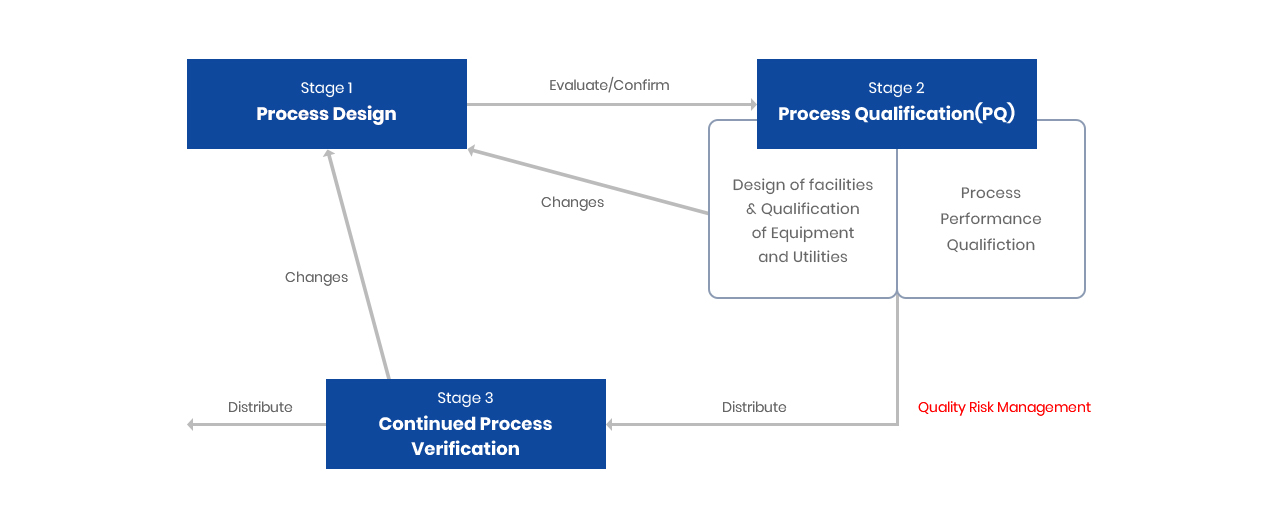
Process Validation Flowchart
 Please move the screen left and right.
Please move the screen left and right.